16 Ago Coral Diseases
The Caribbean has been described as a coral disease “hot spot.” In Puerto Rico, diseases are one of the leading threats to coral health. The impact of disease on corals can be detrimental, leading to large-scale losses in coral cover, diversity, reproductivity and overall reef structure. Coral diseases can occur for a number of reasons, from biotic (e.g. infection, genetics) to abiotic (e.g. temperature stress, contamination) factors. Characterizing the cause of a disease remains a challenge, and disease diagnoses often rely upon visual cues that can vary across time, region, observers, and coral species. Despite these challenges, collaborative research efforts across the Caribbean have improved our understanding and identification of various coral diseases. Below you will find descriptions of some of the more prominent coral diseases in Puerto Rico. The devastating impact these diseases have on reef health highlights the importance of our continued efforts to monitor, research, and restore Puerto Rico’s vital coral ecosystems, complemented with educational programs and actions aimed at restoring healthy environmental conditions.
Coral Diseases in Puerto Rico
Black band disease (BBD), first recorded in Puerto Rico in 1972, was the first biotic coral disease identified in the Caribbean.1,2 It is characterized by the presence of a black band about 0.5 – 3 cm in width that kills coral tissue as it moves across a colony exposing bare coral skeleton.3–5 BBD is a highly virulent disease that affects 25 species of hard and soft corals.6 It is caused by a consortium of a cyanobacteria that includes a sulfide-oxidizing bacteria and a sulfide-reducing bacteria, among others.3,4,7–11 Research has identified Roseofilum reptotaenium as the dominant pathogen,12 although numerous additional bacteria have been found in association with the BBD bacterial consortium.3,9,12–16 Research has also identified that microcystin and sulfide produced by the bacterial consortium facilitate the lethality of this disease, possibly impacting the coral’s symbiotic algae.10,11,13,16 BBD is transmitted through water and direct contact, with corallivores possibly acting as additional vectors.3,17–21 BBD kills coral tissues at a rate of around 0.3-1 cm per day, and multiple studies have found disease prevalence or progression to increase with higher water temperatures, increased light, and nutrient enrichment form land-based pollution sources.3,5,29,18,22–28 Researchers have had success halting disease progression by shading diseased corals, as well as by aspirating and then placing clay or epoxy putty over the black band.30 Experiments have also found that re-introducing the herbivorous sea urchin Diadema antillarum can reduce black band disease progression by controlling competitive algae populations that could otherwise compromise coral health.31
White band disease (WBD) is characterized by a white band of bleached diseased tissue, or tissue-denuded skeleton, that progresses rapidly (0.5 – 2 cm/day) from the base or mid-branches of Acroporid corals.1,2 Two types of WBD (type I and type II), were originally described for the key reef building Acropora species complex, known commonly as Staghorn (Acropora cervicornis) and Elkhorn (Acropora palmata) corals, as well as a hybrid of these two species (Acropora prolifera).1,3–5 These two types were found to be the same disease after more field observations and lab tests were done.6 WBD outbreaks extending from the 1980s to early 2000s coincided with other key threats, including hurricanes, thermal anomalies, and a disease outbreak targeting the key herbivorous sea urchin, Diadema antillarum. These combined threats resulted in unprecedented mortalities of Acropora populations, as well as long-term ecosystem-wide alterations in reef structure and functioning throughout the Caribbean and a shift from coral- to algal-dominance.4,7,8 Nevertheless, signs of genetic resistance and localized population recoveries have been recorded in the Caribbean, including La Parguera in Puerto Rico.9,10 The etiology of WBD is not fully understood, although microbial analyses and the effectiveness of certain antibiotic treatments indicate that bacterial pathogens are responsible.1,11–16 Koch’s postulates (the accepted standard of confirming a causative link between a microbe and a disease) were fulfilled in Puerto Rico, identifying Vibrio charcariae as the main pathogen.6 WBD is transmissible via water, direct contact and animal vectors, including sea snails, damsel fish and plankton.17,18 Spatial analyses have linked increasing sea surface temperature with disease prevalence, indicating that climate change could be a driver of the disease.19–21 Experimental antibiotic treatments of ampicillin and metronidazole have been found to reduce rates of transmission and progression, respectively.13,14 Researchers have also had success halting disease progression by aspirating and then placing clay or epoxy putty over the band.22
White pox disease (WPX), also referred to as patchy necrosis, white patch disease, and Acroporid serratiosis, is characterized by the presence of irregularly shaped white patches of tissue loss on Elkhorn corals (Acropora Palmata).1,2 The patches range greatly in size (2 to 80 cm2) and can develop simultaneously along the colony surface.1 Rates of tissue loss have been recorded at an average of 2.5 – 3 cm/day, with the highest rates occurring in warmer seasons.2,3 In Puerto Rico, outbreaks were first reported in the mid-1990s through the early 2000s in the southwest and Mona Island, with regional prevalence rates ranging from 35 – 74% and 5 – 27%, respectively.4–6 Devastating outbreaks were also recorded in the Florida Keys during this timeframe, where declines of live Elkhorn coral cover reached an average of 88%.1 During this outbreak, the putative pathogen was discovered to be the bacteria Serratia marcescens, after which the name Acroporid serratiosis was developed to refer to WPX cases caused by this pathogen.1,3,7,8 S. marcescens is a bacterial species also found in the human and sheep gut, and research has provided evidence that sewage waste disposal could be a source of the pathogen causing WPX.1,7 Studies since have found that WPX signs have occurred in cases where Serratia marcescens could not be identified as the primary pathogen or even a pathogen at all.9,10 These findings indicate that WPX symptoms can be caused by varying pathogens or stressors, and that the etiology may shift over time and vary by geographic region.9–11 There are somewhat contradicting findings on the mode of WPX transmission; one study in the Florida Keys found that breakout patterns follow a contagious model where infected colonies spread to neighboring corals,3 while another study in the Virgin Islands suggests that high ocean temperatures cause anomalous outbreaks that selectively infect larger and more genetically susceptible colonies.12 There is no known treatment for WPX, however given that the pathogen is a bacterium, it is possible that an antibiotic treatment might be effective at controlling the disease.
White plague disease (WPD) has two variants, type I (WPD-I) and type II (WPD-II), both of which were first described in Florida in the 1970s. Compared with WPD-I which predominantly affects Mycetophyllia ferox and the Orbicella spp. complex, WPD-II has a higher virulence and affects more species (it is estimated that over 40 species have been impacted, Montastraea and Orbicella species being the most prominently affected).1–4 WPD is characterized by rapidly progressing rates of tissue mortality (1-10 cm/day) that leave behind exposed coral skeleton, usually starting at the colony base and moving upwards.2,5 WPD was first detected in Puerto Rico in La Parguera in 1995 and has since been observed throughout the archipelago, including in mesophotic reefs.6,7 WPD prevalence and subsequent coral loss greatly increased in Puerto Rico and the wider Caribbean in 2005 and 2010 in correlation with the two highest thermal anomalies in recorded history, which also induced the two most extensive and intensive bleaching events.6,8–11 This event led to massive declines in key reef building species, which was further exacerbated by bleaching, other coral diseases, hurricanes, and other impacts.6,8,12 This epizootic had long-term ecosystem-wide impacts on coral communities, including a loss of large reef building species.12–15 The etiology of all WPD types is unknown and therefore precise diagnosis is difficult, especially given visual similarities with other white syndromes that also do not have an identified putative pathogen. Multiple studies have found microbial shifts between healthy and WPD affected coral tissue, indicating a bacterial pathogen could play a causative role.16–19 Early research isolated Aurantimonas coralicida as the pathogen of WPD-II,20 however research since has taken WPD samples completely absent of this bacterium and there is no current consensus on a single pathogen associated with WPD symptoms.16–19 Viruses have also been considered as a possible causative agent,21 and certain environmental factors may also play a role in WBD prevalence, including water depth, temperature, light intensity, season, water turbidity, salinity, and levels of oxygen.6,10,22–24 WPD susceptibility varies significantly by coral species, possibly as a result of varying thresholds for microbial dysbiosis, or a microbial imbalance in the coral host that can result in disease.25,26 WPD is highly transmissible and known to spread through water, direct contact, and animal vectors, including the corallivorous worm Hermodice carunculate and the corallivorous snail Coralliophila abbreviate.1,15,27 While treatment is not widely used, research has found phage therapy,28,29 shading23 and epoxy putty30 to be effective techniques at reducing WPD progression.
Aspergillosis (ASP), first recorded in 1996, is a fungal infection that degrades the tissue of several octocoral species.1,2 This disease has lead to significant mortalities in octocorals, with Gorgonian species being the most impacted in Puerto Rico.1,3–5 As the disease progresses (~0.5 cm/month), the skeleton becomes exposed and colonized primarily by algae, and sometimes cyanobacteria, sponges, and other organisms.1,4,6 The putative pathogen has been identified as Aspergillus sydowii, although recent research suggests that other fungal species and bacteria may also play a role in octocoral infections with apparent ASP symptoms.7–9 ASP characterizations vary greatly across time and geographical location, with prevalence and mortality rates depending on a number of factors such as habitat type, depth and species.10–14 There is also a link between ASP prevalence and both high water temperatures and high nitrogen levels, indicating anthropogenically induced stressors related to global warming and coastal pollution exacerbate the disease impact.3,10,15–17 Although ASP is still present in many localities, whole colony mortality is rare and prevalence levels have gone down significantly.3
Since 2005, the appearance of Multifocal Purple Spots (MFPS) in Gorgonian species has been observed in Puerto Rican waters and throughout the wider Caribbean.3,18 This condition, characterized by small (1 – 3 mm diameter) circular multifocal purple nodules, has been progressively spreading throughout La Parguera, Puerto Rico and other Caribbean localities for over a decade.3,19–21 Data suggests the disease is transmitted through direct contact or through water over short distances, and that prevalence is exacerbated by warming water temperatures.3 Dissections conducted on the purple nodules showed that they were produced by a parasitic copepod and that many MFPS infected colonies also harbored the protozoan labyrinthulomycota.3,18,20,21 There is no known treatment for either ASP or MFPS.
Caribbean Yellow band disease (CYBD) is a bacterial disease affecting up to 12 coral species in nearly every region of the Caribbean. It has been recorded in Puerto Rican waters since 1996 and has resulted in extensive coral tissue loss, especially of key reef building Orbicella species, or star corals.1–4 The disease is characterized by yellow blotches (1-5cm), often multifocal, that turn into rings as coral tissue starts to die in the center.5,6 While the putative pathogen has not been confirmed, several bacterial strains of the genus Vibrio have been identified in association with CYBD.5,7,8 Evidence suggests that the pathogens of CYBD target the zooxanthellae, the symbiotic algae that lives within the coral polyp7–10 and that CYBD is not contagious or transmissible by direct contact or water.11 CYBD leads to multiple adverse effects in corals, including reduced fecundity, impaired metabolic and defense processes, loss of tissue, expulsion of the symbiotic algae, and coral death.7,12,13 Rates of tissue loss due to CYBD are variable, with various datasets showing rates ranging from 5 to 36 cm/year.3,7,14,15 Water temperature, reef depth, season and coral species have all been found to affect the disease prevalence and virulence.9,14,16,17 For example, in Puerto Rico, long-term monitoring at La Parguera and Mona Island showed dramatic increases in CYBD prevalence and virulence following a warm water anomaly and coral bleaching event in 2005.18,19 Findings of the spatial distribution of CYBD have varied across locations, from spread-out in Mexico16 to clustered in Puerto Rico.17 Chiseling a firebreak between CYBD lesions and the remaining healthy coral may be an effective mitigation strategy.20 Researchers have also had success halting disease progression by aspirating and then placing clay or epoxy putty over the band.21
Dark spots disease (DSD), also called dark spots syndrome, is characterized by circular, oblong, or ring-shaped dark spots that can increase in size and sometimes cause tissue mortality, often leaving a depression on the colony surface.1–6 DSD affects primarily Siderastrea siderea (massive starlet corals) and Stephanocoenia intersepta, but has also been observed in Colpophyllia natans, Montastraea and Orbicella species, among others.4,6–11 DSD was first recorded in Colombia in the 1990s and was later observed in the western Atlantic in 2001.3,4 DSD is prominent throughout Puerto Rico and the wider Caribbean,4,6–11 although progression rates are relatively slow compared to other coral diseases and whole colony mortality is rare.8,12–14 Some research has even observed hopeful rates of tissue regeneration and colony recovery, although this depends on the region and coral host species.13–15 The etiology of DSD remains unknown. Some research has found evidence that DSD could be a bacterial infection, possibly with connections to Vibrio species.11,16 However, no pathogen has been identified, and some researchers theorize that observed DSD symptoms could be caused by a non-specific stress response or may even represent multiple diseases clumped under one name.1,3,5,8,11,12 Some possible variants have been described over the years, namely DSD type II, dark bands, and ring disease.3,7,8 DSD prevalence and severity can vary with water depth, temperature and nutrient levels, though the existence and nature of such trends varies across time, location and species.6,10,15,17–19 Spatial analyses have mostly found that DSD follows a contagious mode of transmission,6,7,10,20 and several studies have found that DSD can affect the abundance or diversity of symbiotic algae.1,9 Experimental antimicrobial treatments were used during an outbreak in an aquarium in Curaçao, however these were ineffective and never attempted on wild populations.7 No other known treatment for DSD exists.
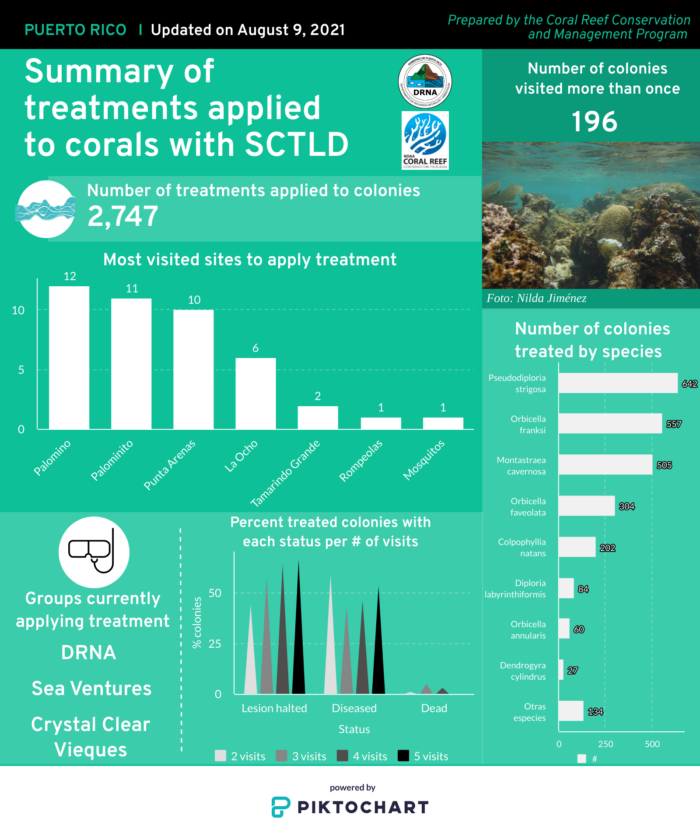
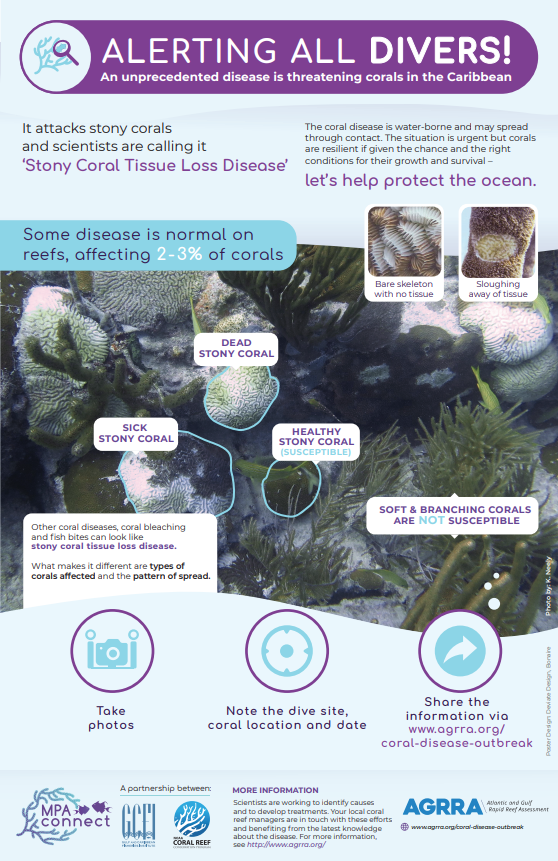
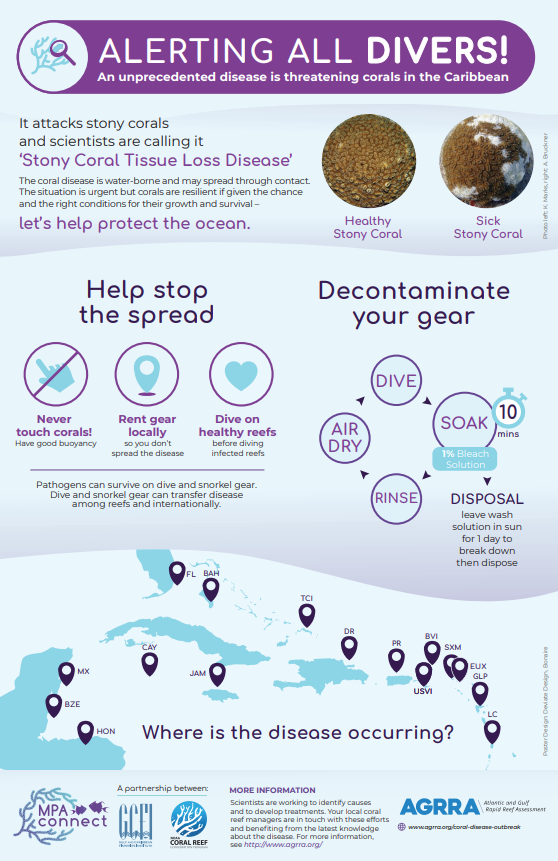
First discovered in Florida in 2014, stony coral tissue loss disease (SCTLD) is a highly virulent, contagious disease known to affect at least 22 species of scleractinian corals.1–3 SCTLD is characterized by focal or multifocal areas of tissue loss that typically progress from the edge of a colony or as white patches within otherwise healthy tissue, leaving white skeleton that becomes quickly (3-7 days) colonized by algae.2,3 This disease has led to unprecedented rates of live coral cover loss across the Caribbean, to the extent that recovery is uncertain and long-term ecological damage is inevitable.2,4–10 SCTLD was first detected in Puerto Rico in Culebra in October 2019. From October to November 2019, surveys in Tamarindo Chico reef showed an increase in SCTLD prevalence from 4% to 50%.11 This fast-moving disease is now widespread throughout Puerto Rico’s eastern, northern and southern coasts, and has even been recorded in mesophotic reef systems (ranging from 23 to 50 meters depth) off of Vieques.12 Due to the high mortality rates and fast spread of this disease, it remains one of the greatest current threats to Puerto Rican reefs. Although the pathogen or pathogens have not yet been identified, both laboratory and field based studies have found an antibiotic treatment of amoxicillin to be effective at halting SCTLD lesions,13–17 providing evidence that bacteria play a role in pathogenesis. Research has also found microbial differentiation between healthy and diseased corals, including significant correlations between SCTLD and the presence of Rhodobacterales and Rhizobiales bacteria, as well as evidence of a possible co-infection related to the bacteria Vibrio coralliilyticus.18–21 Transmission electron microscopy revealed evidence that SCTLD may be related to a viral disease affecting the zooxanthellae (algae symbionts living within coral polyps), which eventually causes coral host death.22 Rapid field-based sequencing techniques developed in the US Virgin Islands identified microbial bioindicators of SCTLD in diseased corals and nearby seawater.23 The relationship between SCTLD and environmental and anthropogenic factors is not fully understood. The disease onset in Florida occurred around the same time as a dredging event and a thermally induced bleaching event, although there is no confirmed link between these events and the disease outbreak.2,4,24–32 Factors that have been found to impact SCTLD manifestation include location, host species, the nature of the lesion, the amount of time the colony has been infected, the amount of remaining live tissue on the colony, the colony size, season, water temperature, and site characteristics such as depth, habitat type, species diversity, coral cover and proximity to a high density of septic tanks.2,3,10,12,26,33–41 SCTLD is contagious and generally spreads through direct contact with diseased tissue or facilitated by medium depth ocean currents.1,18,24,25,39,42,43 It is theorized that ballast water discharge from ships may also explain the rapid and haphazard spread of SCTLD across the Caribbean.10,43,44 Corallivorous fish such as the foureye butterflyfish have been found to predate selectively on SCTLD lesions, possibly impacting transmission.45 Current intervention efforts focus on applying an antibiotic treatment mixed in a specially formulated paste to lesions and rescuing healthy corals for future restoration. Other intervention methods are also being investigated, such as probiotic treatments.42,46,47
Resources for SCTLD
Across the Caribbean, the disease with the current greatest threat to coral reef health is stony coral tissue loss disease. While the causative agent of this disease is not yet known, the high virulence, transmissibility and deadliness of this disease has gotten coral reef conservationists working relentlessly to try and combat this latest severe threat to our reefs. Below are some useful resources to help learn about this disease and ongoing efforts to monitor and respond to outbreaks in Puerto Rico.
This map shows the distribution of sites where Stony Coral Tissue Loss Disease (SCTLD) has been identified in Puerto Rico (shown in red), as well as sites where efforts to treat sick coral colonies are being carried out (yellow stars).
Workshops and Webinars
Documents
- Prospectus SCTLD
- Coral Disease Action Plan
- SCTLD Emergency Response Strategic Plan
- Puerto Rico Stony Coral Tissue Loss Disease Intervention Plan
- Best practice guide for reporting and experimentally processing corals possibly infected with SCTLD - resource in Spanish
- SCTLD Case Definition
- Aquatic census to identify coral colonies with symptoms of Stony Coral Tissue Loss Disease (SCTLD) - resource in Spanish
- List of recommended literature about coral diseases
Useful links
SUBMIT A CORAL DISEASE REPORT

Report sightings of SCTLD to AGRRA (Atlantic and Gulf Rapid Reef Assessment Program)
AGRRA collects and reviews reports of coral disease and bleaching from across the Western Atlantic and Gulf of Mexico. The Department of Natural and Environmental Resources adds these reports to our ongoing database and map of SCTLD distributions in Puerto Rico. This helps us track the disease spread and identify priority sites for treatment and intervention.
Click on either of the following links to enter a Basic or Detailed coral disease or bleaching report. Thank you for helping to monitor Puerto Rico’s reefs!
This simple survey form allows you to enter presence/absence data on corals affected by SCTLD and bleaching by coral species and estimates of corals affected, photos and site information. You can identify survey method used such as rover diver, bar drop, general observation etc.
This detailed survey form allows you to input more detailed quantitative data on the number of coral colonies affected by stony coral tissue loss disease and/or coral bleaching (fully bleached, partly bleached, pale), recent coral mortality, photos and site information. You can identify the method used such as rover diver, bar drop, etc.
Useful AGRRA Resources
VOLUNTEER
Thank you for your interest in volunteering! You can get involved by signing up to be a volunteer for EREA (Equipo de Respuesta a Emergencias de Arrecife, in Spanish).
Please visit EREA’s webpage to learn about how you can sign up. Through this group you will receive information on volunteering opportunities and training sessions for identifying coral species, learning about coral diseases, and treating corals.
The Coral Reef Conservation Program is currently under the Office of Coastal Zone and Climate Change of the Department of Natural and Environmental Resources.
Legal and Historic Background
The Coral Reef Conservation and Management Program was created in response to the Law for the Protection, Conservation, and Management of Coral Reefs in Puerto Rico (Law 147 of July 15, 1999), which empowered the Secretary of the Department of Natural and Environmental Resources (DNER) to develop a program with the particular interest of conserving, managing and protecting these ecosystems. In 2016, the DNER adopted the Coral Reef Regulation (DNER Regulation 8809), with the purpose of providing protection to coral reefs, coral communities and associated ecosystems, regulating it’s uses, establishing penalties that entails their inappropriate use and implementing management mechanisms for their protection and conservation.
At the national level, this Program is part of the Coral Reef Initiative by virtue of Executive Order 13809 promulgated by the President of the United States William J. Clinton, under which the US Coral Reef Task Force. This committee is led by the Department of Commerce (National Oceanic and Atmospheric Administration, NOAA) and the Department of the Interior (DOI), among other federal agencies with inherence on coral reefs. In addition, it is part of the states, territories and associated free states where coral reefs are present.
Management and Conservation Priorities
In 2010, the following management priorities and Local Action Strategies (LAS) were established:
- Improve water quality by reducing pollutants coming from land-based sources.
- Protect reef fisheries and associated ecosystems making use of the management tools available for protection and conservation.
- Reduce human impacts that are most critical to coral health and protection.
- Reduce the impact of phenomena associated with climate change such as diseases that come from the increase in the frequency and impact of storms, the increase in water temperature and air pollution.
- Promote the recovery of coral reefs impacted by previous events.
NOAA’s Coral Reef Conservation Program (CRCP) adopted it’s Strategic Plan in 2018, setting the following national priorities:
- Increase resilience to climate change
- Improve the sustainability of fisheries
- Reduce sources of land-based pollution
- Restore viable coral populations
Puerto Rico Coral Reefs Conservation Program
Department of Natural and Environmental Resources
Postal Address:
San José Industrial Park
1375 Ave Ponce de León
San Juan PR 00926
Telephone: (787)999-2200 ext.2618
Email: programadearrecifesdecoral@drna.pr.gov



























































Sorry, the comment form is closed at this time.